| ||||
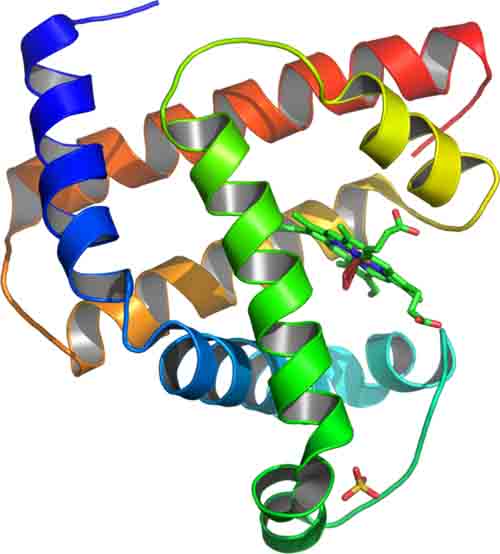
What Are Proteins?
Amino Acids, Peptide Bonds & Levels of Protein Structure
Organic molecules are large chemical compounds that contain carbon-hydrogen bonds and are found in living things. The major classes of organic molecules are carbohydrates, proteins, lipids and
Article Summary: Learn about the chemical bonds and different levels of structure that take amino acid monomers to a whole other level, a complex protein polymer.
What Are Proteins? Amino Acids & Peptide Bonds
 | ||||||
SPO VIRTUAL CLASSROOMS
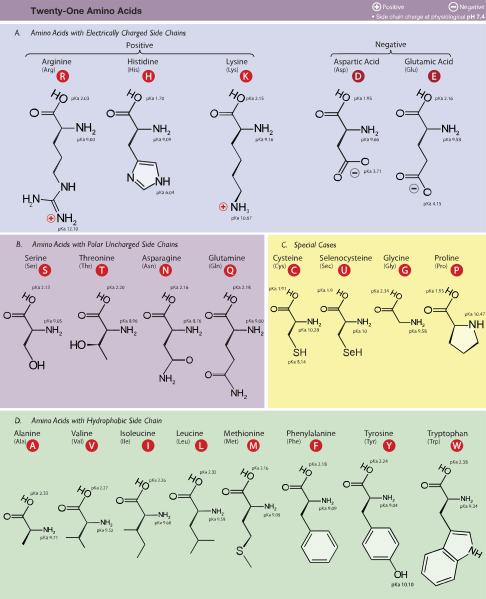
…all attached to same carbon atom (the α–carbon or alpha carbon). A fourth bond attaches α-carbon to a side group that varies among different amino acids. These side groups are important, as they affect the way a protein’s amino acids interact with one another, and how a protein interacts with other molecules.
Although there are hundreds of different amino acids, most organisms use only 21 to build proteins.
Page last updated: 9/2013
The SPO website is best viewed in Google Chrome,
Microsoft Explorer or Apple Safari.
The Virtual Cell Biology Classroom provides a wide range of free educational resources including Power Point Lectures, Study Guides, Review Questions and Practice Test Questions.
Page last updated: 8/2015
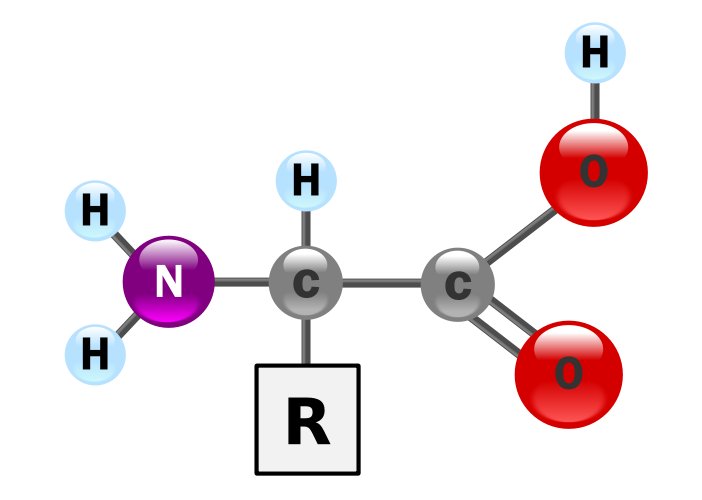
Amino Acid Monomers
Proteins are polymers (big organic molecules) composed of monomers
called amino acids.
Each amino acid contains a…
- base amino group ( -NH2)
- acidic carboxyl group ( -COOH)
- hydrogen atom
Amino Acid Chemical Structure
 | ||||||
SPO is a FREE science education website. Donations are key in helping us provide this resource with fewer ads.
Please help!
(This donation link uses PayPal on a secure connection.)