| ||||
Enzyme Class Experiment:
Catechol Oxidase & Accumulation of Polyphenol in Apples
(How to Prevent Apples From Browning)
CLASS NOTES from Science Prof Online
An enzyme is primarily made of protein (amino acids linked by peptide bonds), but also contains a cofactor—a non-protein portion that is required for the enzyme to work properly.
In summary, enzymes are…
- catalysts that speed up cellular reactions
- not permanently changed in reactions they mediate
- specific to what they catalyze
- reusable
- usually named with an –ase suffix
Article Summary: This simple experiment requires no special equipment and teaches students about enzymes with a tangible example; the browning of an apple due to the enzyme catechol oxidase.
Catecholase Enzyme and Polyphenol in Apples
You have free access to a large collection of materials used in a college-level introductory Cell Biology Course. The Virtual Cell Biology Classroom provides a wide range of free educational resources including Power Point Lectures, Study Guides, Review Questions and Practice Test Questions.
SPO VIRTUAL CLASSROOMS
 | ||||||
 | ||||||
SPO is a FREE science education website. Donations are key in helping us provide this resource with fewer ads.
Please help!
(This donation link uses PayPal on a secure connection.)
Page last updated 10/2014
How Enzymes Work
Unlike inorganic catalysts, enzymes are very specific organic molecules. Each type of enzyme acts on only one particular compound, known as its substrate. The substrate briefly binds with the enzyme, and, in the process, is changed.
Each enzyme has a unique three dimensional shape, including a surface groove called an active site. The active site fits its target substrate much like a key fits in a lock. Other substances that don’t fit can't enter the active site, so no reaction occurs. If the shape of an enzyme’s active site is altered, the enzyme can no longer work, and therefore is considered to be denatured.
How to Prevent Fruits & Vegetables From Browning
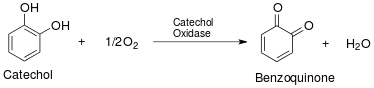
Catechol oxidase (also known as catecholase) is an enzyme present in most fruits and vegetables. It facilitates the browning of cut or bruised produce by catalyzing a reaction between the substrate molecule catechol and atmospheric oxygen (O2). The ultimate product of this oxidation reaction is polyphenol, a brown compound that accumulates when fruits and vegetables are exposed to air. This color change is especially apparent in produce that has white flesh, such as apples and potatoes.
Enzymes are biological catalysts; proteins that help speed up the chemical reactions necessary for life.
Molecular structure of catechol, a colorless phenol.
There are a couple of ways to prevent this enzymatic reaction from occurring. One is to submerge the fruit or vegetable in water. Water doesn’t affect the enzyme catechol oxidase, but does reduce the availability of oxygen, a reactant required for the formation of polyphenol. This is why putting cut fresh potatoes in water delays browning.
Another way to prevent this enzymatic reaction is to remove the enzymes cofactor. A cofacter is the non-protein portion of an enzyme that is required for the enzyme to work. The cofactor of catechol oxidase is a copper ion, which can be removed by changing the pH surrounding the enzyme. When the copper cofactor is removed from catechol oxidase, the enzyme no longer works.
Easy Enzyme Experiment Using Apples
This experiment is so simple that it can be done in any classroom, and requires no special scientific equipment. The supplies for each student include one of each of the following:
- apple
- lemon wedge
- paper plate
- napkin
1. Have each student take a bite of apple, and then immediately rub juice from the lemon over the exposed flesh. Be careful not to let the juice touch other areas of the apple.
2. Each student then takes another bite, on the opposite side of the apple. Do not put any lemon juice on this area of the apple.
3. While the reaction is occurring, the teacher can explain what enzymes are, how they work, etc.
4. After at least a half hour has passed, have the students compare the two bite marks on the apple.
The apple flesh that had been exposed to lemon juice should still be bright white, while the other bite mark, that was not exposed to lemon juice, will have browned. Browning is prevented with lemon juice because its low pH causes the copper cofactor to separate from the catechol oxidase enzyme, after which the enzyme will no longer work. The low pH of citric and ascorbic acids is what prevents the flesh of citrus fruit from browning.
Sources and Helpful Enzyme Links
- Campbell, NA and Reece JB (2005) Biology. Seventh Edition.Pearson Education, Inc.
- Thorpe, P. ed. (2007) Biology 120 General Biology I: Laboratory Experiments & Exercises. Grand Valley State University.
- Enzyme Lecture Main Page from the Virtual Cell Biology Classroom.
- Science Demonstration of the Enzyme Bromelain: Why Fresh Pineapple Cannot Be Used in Jell-O, experiment from SPO.
- How Enzymes Work animation from McGraw-Hill.
The intermediate product benzoquinone continues to spontaneously react to ultimately form the brown product polyphenol.
The SPO website is best viewed in Google Chrome or Apple Safari.
- Interactive Enzyme Tutorial and quiz from Life: The Science of Biology.
- “The Role Enzymes Play in the Body” from Enzymedica. These guys are selling supplements. I don’t endorse the supplement, but their video is very instructive regarding enzymes and digestion.
- “Assignment Discovery: Enzyme Catalysts” from HowStuffWorks, a Discovery Company.
- Activation Energy animation from Pearson Prentice Hall.
- The Protein Song - song by MABS Music
 | ||||
FREE study questions to help students better understand Enzymes: