| ||||
Electron Transport Chain
& Cellular Respiration
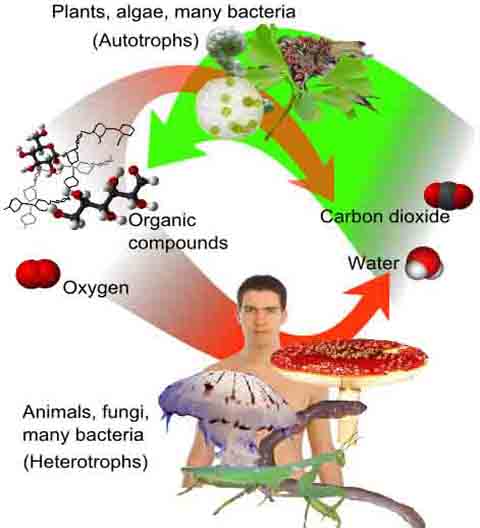
All living things run on energy. If the organism is a plant or autotrophic microbe, the energy comes from sunlight. For all other forms of life, energy is extracted from nutrients through the reactions of metabolism--cellular respiration.
Article Summary: The electron transport chain is the most complex and productive pathway of cellular respiration. Here's a straightforward, simplified explanation of how the ETC works.
The Basics of the Electron Transport Chain
You have free access to a large collection of materials used in a college-level introductory Cell Biology Course. The Virtual Cell Biology Classroom provides a wide range of free educational resources including Power Point Lectures, Study Guides, Review Questions and Practice Test Questions.
 | ||||
See the
for a fun, hands-on way to teach students the steps of the ETC!
 | ||||||
SPO VIRTUAL CLASSROOMS
Cellular Respiration & the Electron Transport Chain
Regardless of whether the original form of energy is sunlight or food, it must ultimately be converted to the cellular energy currency of adenosine triphosphate (ATP).
See Page 3 for an excellent animated video lesson on the Electron Transport Chain!
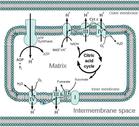
Where the Electron Transport Chain Is Located
Electron transport requires a membrane in order to work. In prokaryotic cells, those of bacteria and bacteria-like Archaeans, electron transport takes place in the cell’s plasma membrane, in folded areas called mesosomes.
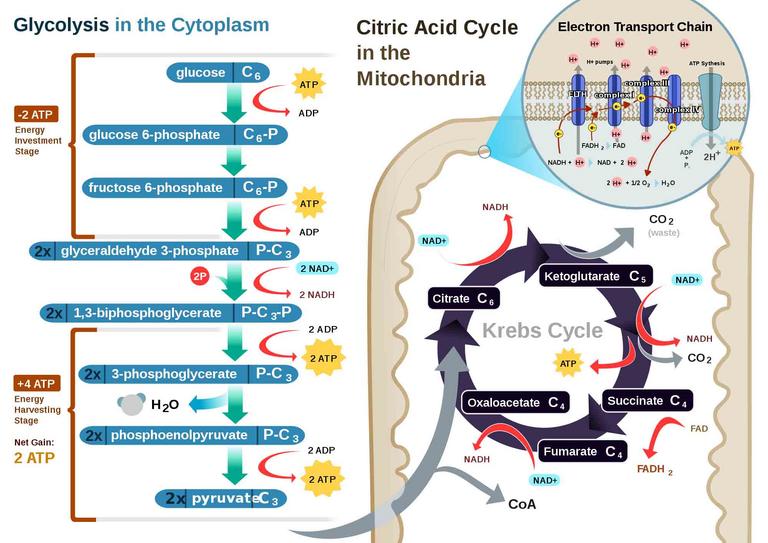
For most organisms, this conversion is accomplished though cellular respiration, a series of biochemical pathways in which glucose (a sugar) is broken down and the energy extracted is converted to ATP.
The pathways of cellular respiration include: 1. glycolysis,
2. conversion of acetyl-CoA, 3. Kreb’s cycle & 4. electron transport. Electron transport is the most complex and productive pathway of cellular respiration. During aerobic respiration, the ETC produces 34 of the 38 ATP molecules obtained from every molecule of glucose.
Page last updated: 3/2016
Adenosine triphosphate (ATP) is the chemical energy that biological cells use to get things done. The energy in APT is stored in its phosphate bonds. ATP is like a rechargeable battery. When it loses it's charge, it becomes ADP (adenosine diphosphate).
Want To Learn More About Metabolism and the Electron Transport Chain?
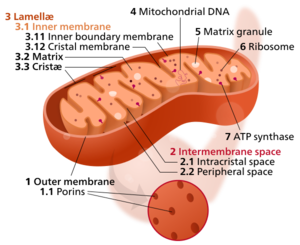
In eukaryotic cells, the more evolutionarily advanced and complex cells of animals, plants and fungi, electron transport takes place in cellular organelles known as mitochondria, where these tiny eukaryotic power factories break down food to make ATP.
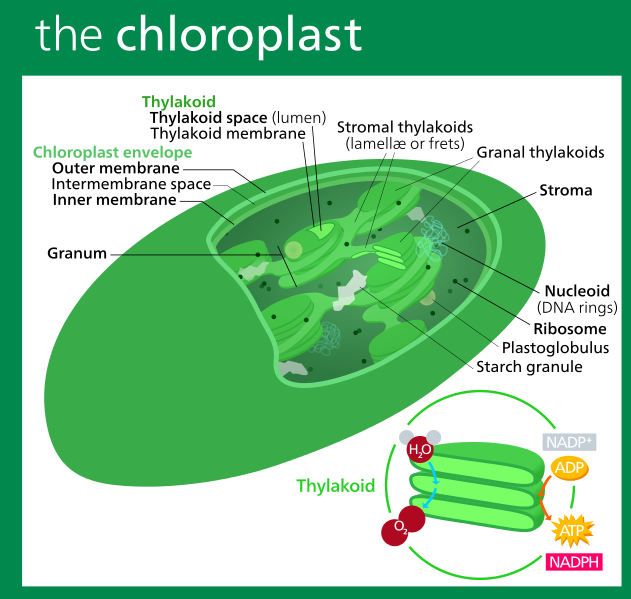
In addition to having mitochondria to turn food into ATP, plant cells also have organelles called chloroplasts with an internal thylakoid membrane, where electron transport chains use sunlight to make ATP.
Whichever type of membrane houses the cell's electron transport chains (plasma membrane, inner membrane of mitochondria or the tylakoid membrane of chloroplasts) cells have many ETCs running continuously, to produce the ATP energy required for cells to survive and thrive.